Introduction
Alzheimer’s disease (AD) is a neurodegenerative disorder primarily associated with amnestic cognitive impairment in its typical form and non-amnestic symptoms in less common variants. It is the most frequent (though not the sole) cause of cognitive impairment in middle-aged and older adults. Its clinical presentation is shaped by specific neurodegenerative and cerebrovascular dysfunctions. In AD, the brain exhibits extracellular deposits of -amyloid (A ) plaques and intracellular neurofibrillary tangles composed of abnormally twisted fibres containing tau protein [1]. AD is thus linked to brain dysfunction resulting from the loss of synaptic homeostasis and impaired metabolic pathways that prevent the formation of A deposits. Research has confirmed that this dysfunction arises from excessive precursor concentrations of A and the aggregation of post-translationally modified tau protein molecules [2–3]. In one study, based on the analysis of correlations between the expression of potential biomarkers and immune system cells, the decrease of RBM3 (RNA Binding Motif 3-biomarker) in astrocytes and the decreased presence of CD8 T cells (Cluster of Differentiation 8-trans-membrane glycoprotein and co-receptor found on the surface of cytotoxic T cells) infiltrating brain tissue were shown to be potential risk factors for Alzheimer’s disease [4]. AD is considered a progressive and incurable disease. Preventative strategies and slowing disease progression depend significantly on health-promoting lifestyle changes, including regular physical activity, diet, vitamin D supplementation, and calcium level monitoring [4–14]. An important intervention is the establishment of support groups and institutional care at various stages of disease development [15, 16]. Equally crucial is the development of programmes and support groups tailored for individuals with AD, whether living independently or under caregiver supervision, in their existing home environment [16–18]. Physical activity and exercises targeting cognitive functions play a pivotal role within these interventions [19, 20].
The aim of this study was to assess the effectiveness of a 24-week standardised exercise programme conducted three times per week for 45 minutes for AD patients, based on the Mini-Mental State Examination (MMSE) questionnaire with the Mungas correction.
Subjects and methods
Participants
The participants of the 24-week exercise programme (i.e., 72 sessions of 45 minutes each) were members of the Memory Problems Support Group Association ‘Niezapominajka’, based in Wroclaw (residents of Wroclaw who remained in their current place of residence), diagnosed and referred by a neurologist or psychiatrist. The exercise programme was conducted from October 2023 to March 2024. Inclusion criteria required membership in the association, written voluntary consent from the participant or their legal guardian The inclusion criteria did not account for the duration of Alzheimer’s disease or the patients’ stage of the disease, despite the relevance of these factors to cognitive function. Medical interviews revealed the presence of various comorbidities among participants, including degenerative changes in the spine and peripheral joints, rheumatic diseases, cancers in remission, pharmacologically controlled hypertension, chronic heart disease, and pain in the shoulders, knees, hips, and spine. Half of the participants had been attending classes organised by the association for more than ten years, while the other half had participated regularly for three to five years.
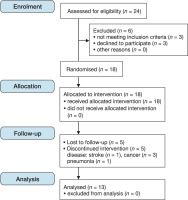
Participants were informed of their right to withdraw from the study at any stage. All patients of the association (n = 24) were offered the opportunity to take part in the programme. For safety reasons, they were divided into two groups of 12 participants each. Due to health-related issues (e.g., flu, common cold, planned medical procedures) or lack of consent, six individuals from one group were excluded at the outset. During the programme, an additional five participants from the second group withdrew due to frequent absences or personal – mainly health-related – issues (Figure 1). Ultimately, the programme’s effectiveness was evaluated based on 13 patients (77% females, n = 10; 23% males, n = 3), aged between 71 and 86 years (mean age 78 ± 6.5) (Table 1). The participants remained in their local environments, living either independently (5 individuals) or under the care of family members or social caregivers (8 individuals). All participants were retirees, with 69.2% (n = 9) having completed secondary education, and the remainder having completed higher or partially higher education. The participants reported various leisure activities, although not all provided detailed descriptions. Most commonly, they mentioned walking, watching television, listening to the radio, and occasionally assisting with meal preparation. The study participants were at different stages of the disease, exhibited varying levels of physical activity, and had diverse comorbidities affecting cognitive function. However, the group was homogeneous in terms of environmental and demographic characteristics.
Table 1
Descriptive characteristics
Measurements
Methods
Anthropometric measurements
Body height (cm) and weight (kg) were measured without footwear using a Zalimp WB-150 medical scale equipped with a telescopic stadiometer. The body mass index (BMI) was calculated (kg/m2) (Table 1) by dividing body weight by the square of the height and categorised according to standard norms: normal weight (18.5 BMI < 25), overweight (25 BMI < 30), and obesity (BMI 30) [21, 22]. In the study group, 23% (3 females) exhibited BMI values indicative of underweight.
Questionnaires
Cognitive function was assessed using the Mini-Mental State Examination (MMSE), which is recommended as a screening tool for cognitive dysfunction (mainly in brain-related dementias) and for monitoring disease progression [23, 24]. Raw MMSE scores were adjusted using the Mungas correction, accounting for age and years of education [25]. Patient diagnostics also included the Clock Drawing Test (CDT) [26, 27].
Exercise procedure
The six-month exercise programme utilised a combination of aerobic and resistance exercises [13, 19]. Sessions were conducted three times a week for 45 minutes each. Under the supervision of a physiotherapist, the exercise routine included activities performed while seated, standing, and walking (e.g., hallway walking). Each 45-minute session incorporated balance exercises (e.g., standing on one leg), coordination tasks (e.g., left hand with right leg), strength/resistance exercises (using bands), assisted exercises (using sticks, balls, and beanbags), anti-thrombotic exercises (targeting ankle joints), and isometric exercises (gluteus muscles, quadriceps) according to geriatric recommendations [24, 28–32]. Exercises were performed both individually and in pairs. Each exercise incorporated mental training elements, such as counting repetitions from 1 to 10 (ascending and descending) which means that each exercise was repeated 20 times. The exercise structure included numerical and verbal components, involving colour recognition, remembering partners’ names, and recalling words beginning with a specific letter. Various sensory-motor tools (balls, bands, sticks, textured balls, balloons, beanbags) were used to provide additional sensory stimulation and variety. Movement tempo and flow were guided by folk music, integrating auditory, tactile, and kinaesthetic stimuli to enhance the therapeutic effects, particularly for older adults [20].
Breathing exercises were performed at the beginning, during, and at the end of the sessions. Typically conducted while standing, these involved a short nasal inhalation accompanied by chest elevation and a prolonged exhalation through the mouth, combined with coordinated arm movements: raising the arms during inhalation and lowering them slowly during exhalation.
Movements involved all joints of the upper and lower limbs, as well as the head and torso. A physiotherapist supervised proper execution and movement tempo. The exercise set followed recommendations that older adults engage in diverse exercises, including endurance, balance, resistance, coordination, and strength training [20, 32–34]. The exercise routine was tailored to the needs and capabilities of older adults and followed the latest physical activity guidelines [20]. The exercises were individually adapted to each participant’s abilities – for example, adjusting the starting position (e.g., performing exercises in a seated position if standing was not possible), an appropriate number of breathing exercises, and rest breaks during the sessions. These exercises promote the ability to perform daily activities independently (e.g., walking, grasping, maintaining balance, and preventing falls) [20, 24, 30, 33, 34].
The physical exercise sessions presented in this study constituted an additional activity incorporated into the regular schedule offered by the association. The patients attended the association’s open day centre three times per week, participating in a variety of activities lasting three clock hours. One group of twelve participants attended the morning session starting at 10:00 a.m., while a second group of equal size arrived at 1:00 p.m., immediately after the end of the first session. Activities were conducted by two qualified occupational therapists, both of whom were professionally trained as nurses and medical vocational instructors.
The physical exercise programme was conducted prior to the main activities of the day. This study’s participants arrived at the association approximately 45 minutes earlier than usual to take part in the exercise session.
The physical activities presented in this study were part of the association’s annual programme. Sessions were organised systematically three times a week, for three hours each, in two groups (2 × 12 patients). For a period of six months, the daily schedule at the association began with the physical exercise session, followed by fine motor activities (e.g., clay modelling, paper crafting, beading, elastic bands), memory exercises (using computer programs, crosswords, puzzles, storytelling, rhymes, brain teasers, colouring books), and educational classes covering disease-specific information, preventive examinations, dietary models, meal preparation, and selection of meal components. Social competence education was also provided [35]. A new element introduced in this 24-week programme was the integration of physical exercises with mental training for members of the association. In conclusion, each participant completed 72 exercise sessions, each lasting 45 minutes.
Statistical analysis
The data analysis was conducted using the Statistica v.13.3 software (TIBCO Software Inc., Palo Alto, CA, USA). The normality of the distribution of quantitative variables (age, BMI) was assessed using the Shapiro–Wilk test. The significance of the differences between the means of the variables with a normal distribution and homogeneous variances was determined using Student’s t-test. The homogeneity of variances was verified using Bartlett’s and Levene’s tests. To analyse the significance of the differences in the parameters derived from the MMSE and CDT questionnaires, the Wilcoxon signed-rank test and the Sign test were employed to compare the effectiveness of the intervention before and after the 24-week exercise programme. Statistical significance was considered at p < 0.05. The power of the analysis was calculated using the statistical module of the SigmaPlot software.
Results
The descriptive characteristics of the patient group with AD are presented in Table 1. The group was homogeneous in terms of age, regardless of sex. Analysis of the BMI values before the programme implementation indicated that the BMI was within the normal range (BMI < 25 kg/m2) for the entire group.
In the study group, 23% (3 females) exhibited BMI values indicative of underweight, supporting the hypothesis that high brain pathologies may underlie the BMI decline preceding dementia disorders [36].
MMSE questionnaire
After the 24-week exercise programme, the study group showed a statistically significant improvement in overall cognitive function across all domains (p = 0.007); however, the statistical power was below 80%, which may be due to the small sample size. Improvement was observed in four participants in the ‘Naming and Repetition’ subdomain, while nine maintained the same level. In the ‘Orientation to Time and Place’ subdomain, improvement was noted in eight participants, no change was observed in four, and one participant showed a decline. The analyses were conducted with statistical power below the desired threshold of 80%. However, improvements within specific domains were not statistically significant. It is important to note the small number of participants who completed the full programme cycle. Cognitive functions remained within the same dementia classification level after 24 weeks, though individual participants showed an increase of 2–3 points compared to their pre-programme scores (Table 2). Analysing the specific sections of the questionnaire post-training, positive changes were observed in the domains related to temporal and spatial orientation, object naming, and repetition of those names (Table 2). However, these improvements were not statistically significant, likely due to the small sample size. No improvements were noted in writing, arithmetic, figure drawing, command execution, memory retention, or recall. The presented study involved a small sample size, and score changes were observed in two subdomains: ‘Naming and Repetition’ and ‘Orientation to Time and Place’. This can be considered a success given the limited number of participants, and serves as a motivation to continue the research. The change in the score for the ‘Orientation to Time and Place’ subdomain may be explained by the fact that the exercise sessions were held in the same association facilities, on the same days, and at fixed times. During each session, participants repeatedly recalled the date, time, and location.
Table 2
Changes in specific MMSE questionnaire components before and after training in the study group
During the exercise sessions, the participants repeated words (such as the names of exercise equipment, the colours of the tools, etc.), thereby reinforcing the vocabulary, which likely contributed to the positive score changes observed in the MMSE subdomain related to ‘Naming and Repetition’.
After the training intervention, statistically significant changes were observed in the entire group when considering the total score across all subdomains of the questionnaire, as illustrated in Figure 2.
Clock Drawing Test
A quantitative assessment of performance on the CDT was employed using the 10-point version developed by Sunderland et al. [37], in which the maximum possible score is ten. In this version, a score between ten and six points indicates a generally correct representation of a complete clock, while scores ranging from five to one point reflect impairments in drawing the clock face, circle, and numbers [37].
Participants who achieved the highest scores on the MMSE questionnaire also obtained the maximum score (10 points) on the clock drawing test, representing 23% (n = 3) of the study group. Notably, the lowest MMSE score recorded by one participant corresponded with the lowest CDT score of 1 point. The remaining participants scored as follows on the CDT: 5 points (1 participant), 4 points (2 participants), 3 points (1 participant), and 2 points (4 participants). No changes were observed in the CDT results after the 24-week programme. The analyses were performed with power less than the desired 80%.
Discussion
It is estimated that approximately 55 million people worldwide suffer from AD, and this number is expected to quadruple by 2050 [12, 17, 38]. In Poland, according to the Institute for Health Metrics and Evaluation, in 2019, approximately 1.5% of the population (379,000–500,000 individuals) were affected by Alzheimer’s disease and related disorders [39]. The incidence rate of AD in Poland stands at 212.93 cases per 100,000 people, with more than half of newly diagnosed dementia cases attributed to AD. The risk of developing AD doubles every five years after the age of 65 [17].
Memory loss is often the first symptom noticed by patients and their caregivers. Both everyday memory and long-term memory are impaired in the early stages of the disease. Memory function impairment is individually associated with structural or functional parameters of brain integrity [2]. AD disrupts memory formation from the molecular level to neural networks. Assessing memory loss in patients enables the identification of neural structures involved in the process, such as the default mode network, epigenetic and genetic factors (e.g., ApoE4 status), and evolutionary aspects of cognition [40].
Despite advancements in the early recognition and diagnosis of AD through cerebrospinal fluid biomarkers and hippocampal volume analysis, neuropsychological testing remains the gold standard for diagnosing AD [40, 41]. Although AD has been linked to -amyloid plaques and Tau protein deposits in the brain, leading to progressive cognitive decline, no treatment has yet been developed to restore cognitive health. Both everyday memory and long-term memory are compromised early in the disease course, with memory impairment associated with structural and functional brain integrity [40]. In clinical practice, memory impairment analysis assists in identifying AD subtypes, determining disease progression, and prognosis.
Physical activity plays a vital role in improving patients’ quality of life, and it was a key component of our 24-week programme. This preliminary research demonstrated a statistically significant improvement in cognitive function after 24 weeks (based on the overall MMSE score). However, despite the clinical and demographic homogeneity, the sample size was small, indicating the need to continue the programme with a larger cohort.
Numerous authors have confirmed the beneficial effects of physical activity on the cognitive functions of older adults with various forms of dementia, including Alzheimer’s disease (AD) [20, 42–44]. Similar to this implemented programme, the literature most commonly recommends a comprehensive ‘multimodal interventions’ approach for older populations. Multimodal interventions are a combination of diet, cognitive training, and physical activity that include adopting an active lifestyle [12, 32]. In this programme, systematic physical exercises were combined with cognitive elements specifically designed for individuals with dementia. It is worth highlighting that patients with AD are generally unaware of the connection between physical activity, motor function, and brain function. Nevertheless, many researchers have confirmed this association, emphasising that regular physical activity in older adults positively influences blood circulation in both the trained body parts and the brain. Regular physical activity has been shown to increase cerebral blood flow (CBF) in the cortical and subcortical regions, as well as the hippocampus, while simultaneously reducing -amyloid (A ) deposits [9, 19, 28, 45]. Exercise induces changes in the brain at the anatomical, cellular, and molecular levels, supporting processes such as angiogenesis, neurogenesis, synaptogenesis, and the stimulation of neurotrophic factors that affect learning, memory, and brain plasticity [9, 12, 19, 20]. Other authors have demonstrated increases in both grey and white matter in the prefrontal cortex following six months of physical training [19] and confirmed the effect of regular exercise on increasing hippocampal volume [46]. The positive impact of physical activity on brain function is undeniable [9, 10, 47–49]. Early diagnosis of cognitive impairments and timely initiation of effective training interventions are crucial. However, there is currently no standardised procedure regarding the components and time frames for an effective exercise regimen for AD in the literature. This study is one of the attempts to address this gap. In our programme, we adopted a schedule of 45-minute sessions three times a week. Other authors suggest different routines, such as 30 minutes five times a week, 20 minutes three times a week, or 40 minutes three times a week. The duration of the described interventions also varies, ranging from 12 weeks, 16 weeks, six months, and even up to a year. The effectiveness results differ, potentially due to disease progression and variations in pharmacological support [10, 13, 20, 33, 42, 44, 48].
This heterogeneity of interventions makes it challenging to compare our results with those of other researchers [48], considering the inevitable deterioration of patient health and individual physiological responses.
In this study, we confirmed that cognitive functions in the observed group were maintained at the same level due to the systematic and prolonged nature of the training, which aligns with findings from other studies [20, 29].
During this study, we also observed that patients performed some simple, unilateral exercises correctly due to prior familiarity. However, new movements, particularly alternating exercises that involved crossing the body’s midline and integrating verbal instructions, posed difficulties and required longer training periods to establish movement patterns.
The aerobic exercises applied in this study, conducted three times a week for 45 minutes, positively affected changes in the MMSE questionnaire results. This finding is consistent with a meta-analysis conducted by Zhang et al. [13], which confirmed the significant impact of aerobic exercises performed with the same frequency and duration on improving MMSE scores in AD patients [13]. Our research on the programme’s effectiveness requires continuation with a larger group of patients.
Conclusions
The 24-week physical exercise programme significantly improved overall cognitive function in patients with AD, as assessed by the MMSE questionnaire. Physical exercises combined with cognitive training over 24 weeks yielded therapeutic benefits in maintaining cognitive functions. However, the assessment of the programme’s effectiveness requires further research with larger patient groups, also taking into account the duration of the disease, which may potentially improve the statistical power of the test.
In conclusion, the present study demonstrated the impact of physical activity on the maintenance of cognitive function. The results of the six-month exercise intervention, which was incorporated as an additional activity into the association’s regular programme, are promising. As part of this pilot initiative, the patients engaged in 45-minute physical activity sessions prior to their regular three-hour programme. Although our aim was to involve all patients of the association (twenty-four individuals divided into two groups of twelve) in the exercise component, this proved impossible. Ultimately, thirteen patients participated in the programme.
Further research will involve not only increasing the sample size with respect to demographic and environmental homogeneity but also including a group of AD patients residing in care facilities, as well as a control group of older adults without diagnosed neurodegenerative diseases, for example, participants of the University of the Third Age in a similar age range, who will undergo physical activity sessions.
Limitations
A limitation of the present study was that the study group was heterogeneous in terms of the length of time between diagnosis and programme initiation. Additionally, information regarding the type and duration of pharmacotherapy used by the patients for their primary condition and comorbidities was not obtained. However, the group was homogeneous in terms of age, social conditions, and environment. A positive aspect of the study was the 24-week intervention, adhering to the principles of single health training sessions and the repetition of its elements. During the programme, we also considered individual MMSE component deficits and modified exercises to accommodate patients’ capabilities. An innovative feature of the project was combining movement with verbal expression, which further activated and strengthened patients’ speech abilities. This study should be considered preliminary research for developing a health training model for patients with AD in Polish conditions.